|
12/28/2023 0 Comments Antimony orbital diagram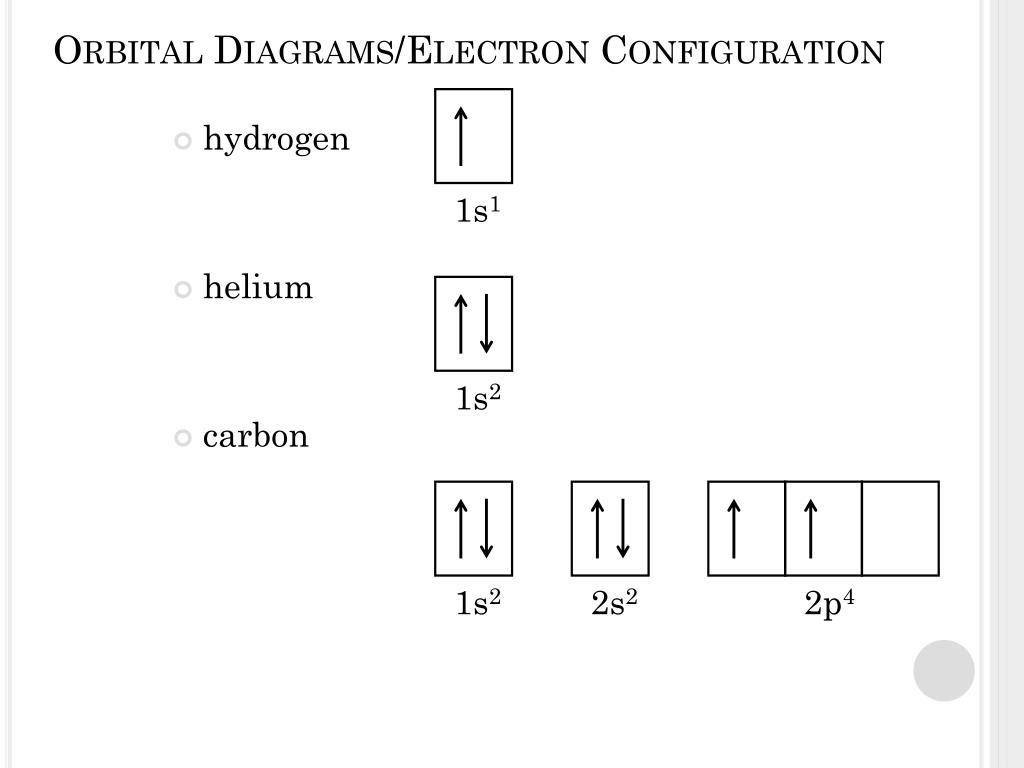
(If you cannot draw arrows, just use u for up arrow and d for down arrow. So each s subshell has one orbital, each p subshell has three orbitals, each d subshell has five orbitals, and each f subshell has seven orbitals.Īs mentioned above, the electron configuration of tin is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 10 5p 2. Give the orbital diagram just for the valence electrons of Antimony atom (symbol: Sb). 
Where, ℓ = azimuthal quantum number of the subshellįor s subshell, ℓ = 0 For p subshell, ℓ = 1 For d subshell, ℓ = 2 For f subshell, ℓ = 3 We can calculate the number of orbitals in each subshell using the formula: 2ℓ + 1 Hund’s rule – each orbital should be first filled with one electron before being paired with a second electronĪlso, you should know the number of orbitals in each subshell.Pauli exclusion principle – two electrons with the same spin can not occupy the same orbital.Aufbau principle – electrons are first filled in lowest energy orbital and then in higher energy orbital Write the complete electron configuration and draw an orbital diagram for antimony.#3 Draw Orbital Diagram of Tinīefore drawing the orbital diagram, you should know the three general rules. Now in the next step, start drawing the orbital diagram for tin. Learn how to find: Tin Electron Configuration motion is in nearly uniform rectilineal directions in liquids, in irregular directions : and in solids, orbital or oscillatory. The electron configuration of tin is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 10 5p 2. Here, the electron configuration of iron ion (Fe 2+) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 6. Since the atomic number of tin is 50, the total electrons of tin are 50. The iron atom donates two electrons in the 4s orbital to form an iron ion (Fe 2+ ). The atomic number of tin represents the total number of electrons of tin.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
